I have just realised that I haven’t written a onein400 research post for over 6 months! Apologies, readers. Let me correct this right now!
This one is split into two distinct sections. First, a quick, but timely, update about the pipeline for new drugs/treatments and then a discussion about genetics and the rather indistinct split between so-called inherited or familial MND and all other cases (Sporadic).
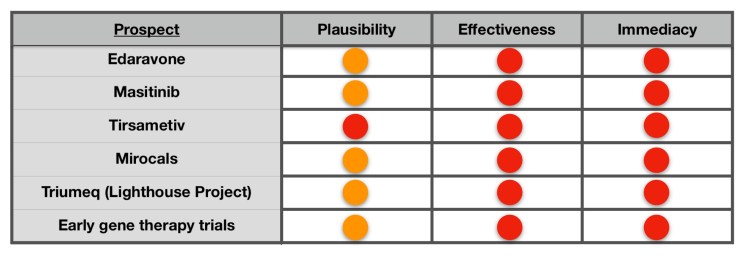
Picking up from previous posts (see my research page for details) here is my view of the drugs/trials that appear closest to some potential realisation. I say potential realisation as opposed to availability, as many drugs can look promising but until the day a drug is approved (for example by NICE and the MHRA in the UK) it is simply not available. Some readers may question my list, stating missing projects. However, I exclude anything that is in such early stages that I see them as some time away from meaningful human trials at this time.
In order to help me, I thought that introducing a colour coded status for my thoughts on any drug/treatment might visually show current status. So, in the good old traditions of traffic lights (red to green) here it is in three parts:
plausibility – does the drug have a plausible mode of potential action?
effectiveness (including patient burden) – how effective has it been currently proven to be? (UK/European trial standards).
and immediacy how far away from being in my pill-box or doctor’s toolbox?
For example, a green, green, green drug would be highly plausible, proven highly effective and just awaiting final approval before patient use. At the other end of the spectrum a red, red, red drug would be no plausible mechanism as yet, no proven effectiveness (UK medicines agency) and, of course not available.
I will say right now and upfront, after looking at my chart below, you will probably think “What that bad, what is going on?” However, I feel this depiction is needed to bring reality to the situation. But there is good news mixed in amongst this current position, and in addition the status is fluid and may change up or down (ie red can change to amber or green as we go forward).
The drugs/treatments are
Edaravone is the new drug approved in the USA just last year (brand name Radicava). It is still unlikely to be approved here in the UK due to more stringent UK and European trial proof levels. There will have to be additional trials for this drug to make any headway in Europe. These do NOT appear to happening yet. My personal thoughts on this drug (in its current formulation) is red for the reasons I have written about before. It has a very small effect (and yet to be proven for UK standards) and a rather nasty burden of the patient being hooked up to an I.V drip every day for 14 days every month! (Being forced to be immobile with a progressive mobility disease!) I recently had an I.V feed in my neck following heart surgery, and the port that was there became excruciatingly painful after just 4 days. It’s a non starter for me.
However, the small effect that has been observed with this drug has sparked a new revolution in analysing groups of patients during trials. Historically, trials have had to show a statistically beneficial response averaged across all patients within the trial. This analysis, and others, are now showing hidden effects in sub-groups within overall participants! In fact this is probably the reason why the EMA will not approve the drug as the effect was found only in the post trial analysis. To remove all bias, a new study would be required with an equivalent sub group in a new trial and in greater numbers. This may happen, but I remain unconvinced because of the patient burden.
The lessons learnt are going into many new drug trials and this is a major advance.
Masitinib is a tyrosine kinase inhibitor that has shown some effectiveness. The data, to me, looks good enough to justify pursuing. The drug was currently being reviewed by NICE in the UK, but on the 28th Feb 2018, the review was suspended because it appears the pharmaceutical company had not submitted required documentation in time for the process to proceed! My personal thoughts of this drug, before the 28th Feb, was that it our best prospect for the second drug to be added to our armoury here in the UK. I am trying to find out what has happened. It still may be the next hope.
Tirasemtiv was a drug aimed at strengthening weakened muscles. It was not intended to be a cure nor a disease halting therapy. Even so, it now has failed phase 3 as the side effects were far too serious. The company is looking at a variation of a similar drug for future trials. So for now, this is off the list.
Mirocals, which I mentioned in my last post has recruited the first patients. This is using low dose Interleukin 2 as a way of dampening the response to any potential immune reaction perhaps caused by MND. Although MND is not considered an auto-immune disease, there is some evidence that once MND takes hold our immune system might exacerbate the disease. This is promising and will take the next 18 months to complete phase 2 trials. My Onein400 rating is definitely Amber on this one.
Triumeq is an anti retro viral therapy drug already used to treat HIV/AIDS. Some studies have implicated endogenous retro virus DNA (which we all possess) in a group of MND patients. Long thought inert, some evidence has indicated that it may not be. Although we don’t know if it is disease causing, an attempt at using a safe and proven anti retro viral drug is a very reasonable trial step. Interestingly this trial will measure some bio markers for MND. This could advance MND trials across the board by speeding up trials (more good news).
I have mentioned gene therapy in previous posts, and there are number of trials underway. One is at multiple centres around the globe, including Sheffield in the UK. I believe early results of Phase 1/2 will be available early 2019. This I find the most exciting of all research going on and that the plausibility of finding a more lasting treatment is amber/green.
Now, before I hear shouts of, ”Hey, Lee! Isn’t there, this, this and this going on?” The answer is “yes”, but we are talking about the majority of other research being pre-clinical. There is no doubt that the rate of progression in research is accelerating.
Please remember that even if a drug was green, green, green here in the UK and the rest of Europe the actual availability will require government regulatory review and price negotiations with the manufacturer.
I now want to return to the title of this post, “One thing leads to another”.
I hope this will complement the above, which some of you might possibly might have found a little disheartening. Let’s look at one aspect of the disease that our understanding of is rapidly evolving and is having profound effects on the search for a therapy.
What is the difference between inherited MND and so-called sporadic MND?
It’s probably best to start this section off with a very pertinent fact. Public understanding of inherited disease is often a simple “I inherited a gene” and therefore will get a disease or not. This can be true for many diseases for which there is a 100% single gene failure. It’s very predictable.
This is simply not the case with MND.
In reality the distinction between inherited MND and sporadic MND is becoming more and more arbitrary by the day and can both confuse and mislead. This short discourse describes why it may not be the best way to describe the disease.
Although about 20 genes have been associated with MND, they are just that, associated. This often gets translated into the general statement that gene x caused MND. Largely it is not even know if these gene mutations are in fact disease causing or simply involved. There is evidence that certain genes affect the speed of disease progression but it can even vary within one gene. The best that can be said is that they constitute a burden for which makes people more likely to develop disease. Importantly it is not possible to say that the sporadic MND cases are not finally manifested by the same or related metabolic transitions. It may be that the burden only requires a simple interaction rather than a more complex process, but still touching similar processes!
To give you a bit more insight into genetics, it’s useful to explain how a gene, or more specifically a mutation, is identified in research to actually be disease associated.
If the genome of an individual with a disease like Huntington’s Chorea, for example, it is found every patient has a particular mutation. It is an absolute fog horn of a marker.
The early MND genetic research that identified a mutation in gene SOD1, took the analysis of many 100s of cases to see the significance. As time progressed, and more and more data has become available, other genes have shown significance. Typically the newer genes/mutations found will be associated in less and less cases. They are only detected through the analysis of more and more cases. This is often known as the “long tail”, where huge amounts of data can show significance which cannot be seen in a few cases. There is even the possibility that some (or even a lot of) MND cases are caused by totally unique mutations, ie ones that will never be able to detected by definition, ie unique!
Currently about 10% of MND cases have some known linkage to genetics. However, it is most likely significantly more, and it is certain that this will increase as we analyse more and more data. In fact, it is actually possible to quantify potentially how many cases may be genetically linked.
Through the analysis of twin studies, where at least one has developed MND, the genetic influence is estimated at about 65%!!
Some new research builds on an article released in 2015. In his paper in 2015, Ammar Al-Chalabi and his team showed that MND is a multi-step disease, ie a number of steps have to occur before the disease manifests. His earlier work showed that there are probably 6 distinct steps involved. The new research, showcased at the 2017 MND Symposium developed the work further, focusing on 2 particular genes involved. Interestingly this data has shown that two steps are required for one gene mutation and three for another gene!! This further supports the “genetic burden” combined with other factors that actually trigger the disease.
It all leaves open approaches to treatment that may actually help identify common pathways that can be interrupted for all patients rather than just genetic or sporadic cases.
The distinction is now getting fuzzy but the search for a therapy is accelerating.
I am going to follow-up this post very shortly with a very brief piece on the topical subject of personalised medicine and MND which is very closely related to the discussion above.